Overview - Clinical Translation Start Trial Program (ClinicStarT)
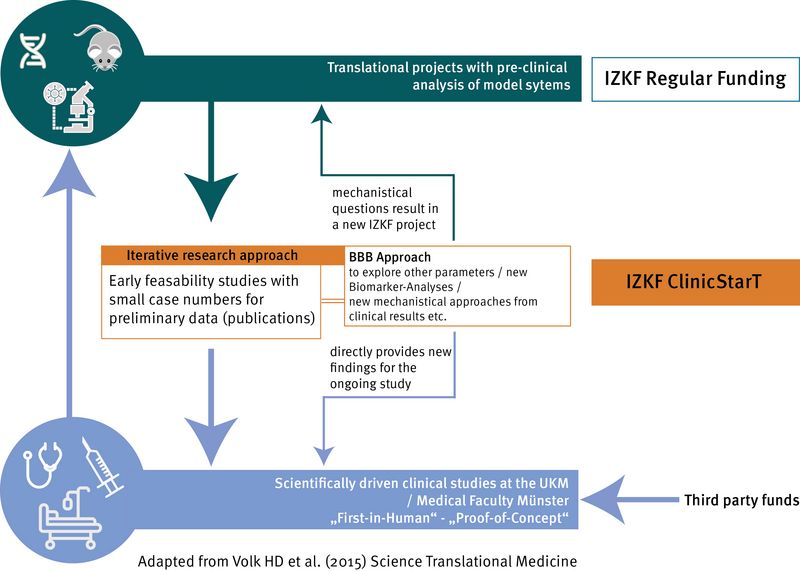
In order to strengthen patient-centred clinical research, the IZKF Münster funds time-limited innovative concepts and science-driven own ideas ("Investigator-initiated trials", IITs) for clinical trials, which are intended to close the funding gap between findings from basic research and patient-centred research and to bring the findings into clinical trials. The fundamental goal of this new IZKF funding format is to increase the implementation of early IITs/pilot studies in the Medical Faculty.


