Institutassoziierte Forschergruppe "Zelladhäsion und Zellpolarität"
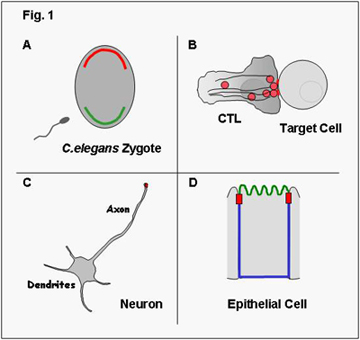
Cell Polarity
Polarization is a universal trait of eukaryotic cells and every cell acquires features of polarization – either transiently or stably – during its life. In some cases cell polarity is reflected by an asymmetric distribution of molecules within the cells and as such is not visible unless these molecules are visualized. For example, in the fertilized egg of C.elegans certain proteins localize exclusively to the anterior pole (shown in red in Fig.1A) and others exclusively to the posterior pole (shown in green). Epithelial cells, as another example, have distinct membrane domains, an apical (shown in green in Fig. 1D) and a basolateral membrane (shown in blue), which differ in the composition of proteins; this is also referred to as membrane polarity. In other cases, polarization reflects morphological asymmetry and is illustrated for example by the morphological changes in cytotoxic T lymphocytes (CTL) during lysis of target cells (Fig.1B) or by the presence of a single axon and several dendrites in neurons (Fig.1C).
Clearly, polarization is required for the cell to function properly. For example, the presence of several dendrites and a single axon in neurons determines the unidirectional flow of signals. Also, the formation of different membrane domains in epithelial cells with distinct identities is required for the directional uptake of nutrients from the lumen of the gut by intestinal epithelial cells. The establishment of cell polarity is thus not only required for the functioning of the individual cell but is a prerequisite for the development of whole organs and consequently the entire organism.
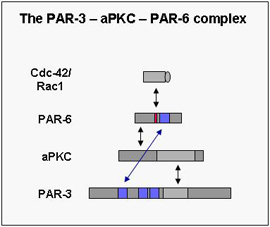
Cell Polarity Proteins
In the last few years the molecular mechanisms underlying cell polarity formation have attracted much interest in cell biology. Several highly conserved proteins have turned out as master regulators of cell polarity. One group of cell polarity proteins consists of PAR proteins (PAR-1 to PAR-6) with a complex consisting of PAR-3, aPKC and PAR-6 forming a functional unit. The PAR proteins have originally been identified in C.elegans in a genetic screen for genes regulating the partitioning of cytoplasmic granules (par stands for partitioning-defective), and PAR-3 and PAR-6 together with atypical PKC form a functional unit (the PAR-3 – aPKC – PAR-6 complex). Meanwhile the PAR proteins have turned out to be used by a large variety of cell types as diverse as fertilized eggs, epithelial cells, neurons, astrocytes or leukocytes, and to be involved in processes like membrane specification, tight junction formation, axon specification or cell migration. In part, the ability to regulate such diverse functions resides in the ability of the PAR-3 – aPKC – PAR-6 complex to influence microtubule polymerization and stability.
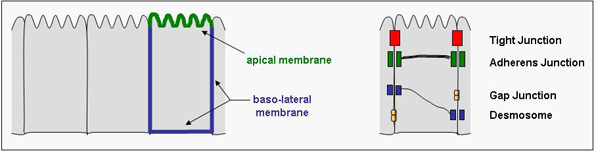
Epithelial and endothelial cells
We have focussed our research interest on the mechanisms underlying the formation of cell polarity in epithelial and endothelial cells of vertebrates. Epithelial cells often form monolayered sheets of cells that line surfaces of organs and form the interface between the interior and the exterior of the organism, for example in the respiratory tract, the gastro-intestinal tract, or the genito-urinary tract. Endothelial cells are specialized epithelial cells that line the inner surface of the blood vessels. In both cell types, the plasma membrane is divided in two domains: the region of the membrane which is "free", i.e. not in contact with another cell, is defined as the apical domain, the membrane regions which are in contact with adjacent cell or with the underlying extracellular matrix are defined as the basolateral domain. These domains are separated by cellular junctions, in particular the tight junctions in vertebrates, which maintain this membrane polarity by preventing the intermixing of the two domains. Understanding the mechanisms regulating the formation of tight junctions is therefore also critical to the understanding of membrane polarity in epithelial and endothelial cells.
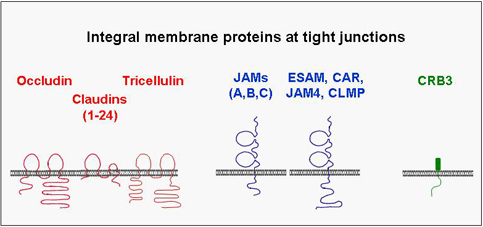
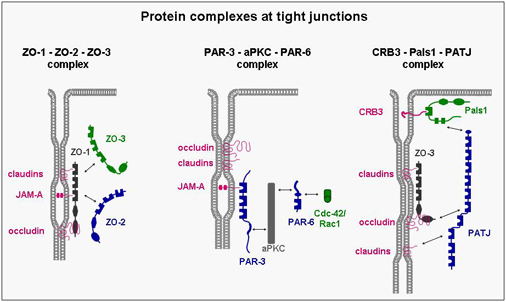
A number of integral membrane proteins has been identified at the tight junction. Many of these mediate adhesion between neighbouring cells through homophilic interactions and help to anchor the individual cell within the multicellular sheet. They all associate with cytoplasmic proteins and are frequently linked through these interactions with the actin cytoskeleton. Although the biological role of these interactions is still unknown in many cases, it became clear that they are far from being static but rather highly dynamic and subject to multiple modes of regulation. One principle that has emerged during the last few years is that the integral membrane proteins through their association with cytoplasmic proteins help to organize large, multimolecular signaling complexes at the tight junction.
Cell adhesion molecules in cell polarity
We are primarily interested in two families of cell adhesion molecules, Junctional Adhesion Molecules (JAMs) and Cadherins, and their role in cell-cell contact and cell polarity formation.
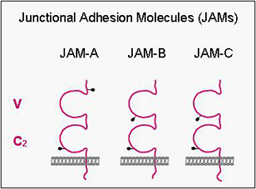
Junctional Adhesion Molecules (JAMs)
JAMs comprise a small subfamily within the immunoglobulin superfamily consisting of three members (JAM-A, -B, -C). They all undergo homophilic interactions and are expressed by different cell types including epithelial cells, endothelial cells, leukocytes and in cells of the reproductive system (Sertoli cells, spermatids). In epithelial and endothelial cells, they are localized at tight junctions. We have focussed our work on the identification of intracellular binding partners of JAMs and have identified several proteins including ZO-1, AF-6/afadin and PAR-3. These proteins have in common one or multiple protein-protein-interaction domains like PDZ domains or guanylate-kinase domains, and they serve to organize the formation of multiprotein complexes. Thus, one major function of JAMs probably resides in the recruitment of specific protein complexes including cell polarity protein complexes to sites of cell-cell adhesion.
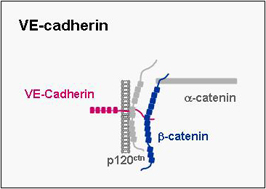
VE-cadherin
VE-cadherin belongs to cadherin superfamily, mediates homphilic binding and is expressed specifically in endothelial cells where it localizes to adherens junctions. It mediates cell-cell adhesion and as in the case of other classical cadherins its cytoplasmic tail binds β-catenin and p120ctn. We have recently identified a cell polarity protein complex in endothelial cells which is associated with VE-cadherin through direct interactions of both PAR-3 and PAR-6 with VE-cadherin. As opposed to the hitherto known PAR protein complex described in epithelial cells, the VE-cadherin-associated PAR complex lacks aPKC. The biological role of this PAR complex in endothelial cells is a major focus of our present work.
Thölmann S, Seebach J, Otani T, Florin L, Schnittler H, Gerke V, Furuse M and Ebnet K (2022). JAM-A interacts with α3β1 integrin and tetraspanins CD151 and CD9 to regulate collective cell migration of polarized epithelial cells. Cell. Mol. Life Sci. Jan 24;79(2):88. doi: 10.1007/s00018-022-04140-5.
In this paper, we have addressed a potential role of JAM-A in the regulation of collective cell migration in polarized epithelial cells. We found that JAM-A depletion slows down collective cell migration, which is most likely caused by a reduced dynamics of cryptic lamellipodia and reduced motility of individual cells embedded in the collective. Again, we identified an interaction of JAM-A with tetraspanins (CD151, CD9) and with a member of the integrin family (α3β1). This is the second example of a close physical association of JAM-A with an integrin that is mediated by a tetraspanin. We interpret our observations in a way that JAM-A teams up with integrins to regulate integrin-mediated functions, and that Tspans serve to physically link JAM-A with integrins to allow a regulatory crosstalk of JAM-A with the integrin.
Haas AJ, Zihni C, Ruppel A, Hartmann C, Ebnet K, Tada M, Balda MS and Matter K. (2020). Interplay between extracellular matrix stiffness and JAM-A regulates mechanical load on ZO-1 and tight junction assembly. Cell Rep. 32(3):107924. doi: 10.1016/j.celrep.2020.107924.
This study performed in the lab of Karl Matter and Maria Balda in London (UK) identifies JAM-A as a regulator of tensile forces acting on tight junctions. The study uses a ZO-1-based tension sensor that senses tensile stress on tight junctions. JAM-A turned out to limit RhoA activity at the tight junctions thereby limiting stress fiber formation and mechanical forces at the tight junction. Surprisingly, the mechanical forces acting on the tight junctions are dependent by the machanical properties of the extracellular matrix. These findings thus also identify JAM-A as signal transducer acting at the interface of cell-matrix and cell-cell adhesion.
Hartmann C, Schwietzer YA, Kummer D, Kirschnick N, Hoppe E, Thüring EM, Glaesner-Ebnet M, Brinkmann F, Gerke V, Reuter S, Nakayama M and Ebnet K (2020). The mitochondrial outer membrane protein SYNJ2BP interacts with the cell adhesion molecule TMIGD1 and can recruit it to mitochondria. BMC Mol Cell Biol. 2020 Apr 17;21(1):30. doi: 10.1186/s12860-020-00274-1.
In this study, we have focussed on a JAM-related adhesion molecule called TMIGD1. Like all JAMs, TMIGD1 contains two Ig-like domains, a single transmembrane domain and a short cytoplasmic tail that ends in a PDZ domain-binding motif. In a yeast-two hybrid screen we identified various binding partners for TMIGD1. This paper describes the characterization of the TMIGD1 interaction with SYNJ2BP, a PDZ domain-containing scaffolding protein that has been found to be localized both at the mitochondrial outer membrane and at the plasma membrane. We observed that TMIGD1 localizes to mitochondria when cells are subconfluent, most likely through its interaction with SYNJ2BP. A localization at mitochondria has not been observed for a JAM-related adhesion molecule before.
Otani T, Nguyen TP, Tokuda S, Sugihara K, Sugawara T, Furuse K, Miura T, Ebnet K and Furuse M (2019). Claudins and JAM-A coordinately regulate tight junction formation and epithelial polarity. J. Cell Biol. 218(10):3372-3396.
The role of JAM-A in the regulation of the barrier function of epithelial cells has not been clear for a long time. This work performed in Mikio Furuse’s lab in Okazaki (Japan) provided evidence that JAM-A is directly involved in the epithelial barrier function. In the absence of claudins, epithelial cells loose their barrier properties for small ions but retain their barrier function for macromolecules. Additional depletion of JAM-A in claudin-deficient cells breaks down the barrier for macromolecules concomitant with a loss of membrane appositions. Through its adhesive function, JAM-A thus directly contributes to the barrier function of epithelial tight junctions. This study provided important insights into the function of JAM-A at tight junctions.
Ebnet K (2017). Junctional adhesion molecules (JAMs): Cell adhesion receptors with pleiotropic functions in cell physiology and development. Physiol. Rev. 97: 1529-1554. doi: 10.1152/physrev.00004.2017.
This review article provides an overview on the physiological functions of the JAM family members JAM-A, JAM-B and JAM-C. It also describes in detail the molecular mechanisms underlying the various functions. It contains a number of illustrations which should allow the interested reader to gain a quick insight into the diversity of JAM functions at the molecular, cellular and tissue level.
Fededa JP, Esk C, Mierzwa B, Stanyte R, Yuan S, Zheng H, Ebnet K, Yan W, Knoblich JA and Gerlich DW (2016). MicroRNA-34/449 controls mitotic spindle orientation during mammalian cortex development. EMBO J. 35: 2386–239.
This study performed in the lab of Daniel Gerlich at the IMBA in Vienna (Austria) provides evidence that JAM-A’s function in regulating mitotic spindle is relevant during development of the cerebral cortex, illustrating one example of the in vivo relevance of JAM-A as reglator of mitotic spindle orientation.
Brinkmann BF, Steinbacher T, Hartmann C, Kummer D, Pajonczyk D, Mirzapourshafiyi F, Nakayama M, Weide T, Gerke V and Ebnet K (2016). VE-cadherin interacts with cell polarity protein Pals1 to regulate vascular lumen formation. Mol.Biol.Cell 27: 2811-2821.
In this paper, we identify another conserved cell polarity protein as interaction partner for VE-cadherin. Pals1 is part of the CRB3 – Pals1 – PATJ complex which localizes at the tight junctions in epithelial cells. We found that Pals1 directly interacts with VE-cadherin further emphasizing that VE-cadherin acts as a regulator of cell polarity in endothelial cells. Our observations also suggest that the differences in the organization of cell-cell junctions between endothelial cells and epithelial cells may in part be explained by VE-cadherin’s ability to organize cell polarity proteins at the adherens junctions.
Tuncay H, Brinkmann BF, Steinbacher T, Schürmann A, Gerke V, Iden S and Ebnet K (2015). JAM-A regulates cortical dynein localization through Cdc42 to control planar spindle orientation during mitosis. Nat Commun. 2015 Aug 26;6:8128. doi: 10.1038/ncomms9128.
In this study, we describe a very unexpected function of JAM-A. By analyzing 3D cyst formation of MDCK cells expressing a dominant-negative mutant of JAM-A we observed that cyst developed multiple lumina instead of a single lumen, a phenotype that was previously observed when cells failed to orient the mitotic spindle apparatus in the plane of the cellular sheet. We found that JAM-A triggers a transient activation of Cdc42 and PI(3)K, generates a gradient of PtdIns(3,4,5)P3 at the cortex, and regulates the formation of the cortical actin cytoskeleton, which are all required to anchor the astral microtubules at cell-cell contact sites and thus to prevent random rotation of the spindle apparatus during cell division. A role in the regulation of mitotic spindle orientation has so far been unique among JAM family members.
Peddibhotla SSD, Brinkmann BF, Kummer D, Tuncay H, Nakayama M, Adams RH, Gerke V and Ebnet K (2013). Tetraspanin CD9 links JAM-A to αvβ3-integrin to mediate bFGF-specific angiogenic signaling. Mol.Biol.Cell 24: 933-944.
In this paper, we describe the identification of the tetraspanin family member CD9 as interaction partner of JAM-A. Tetraspanins are proteins with four membrane-spanning domains and two extracellular loops. They interact with many other proteins including other integral membrane proteins through their extracellular loops, their membrane-spanning regions and their cytoplasmic domains thereby forming so-called tetraspanin-enriched microdomains (TEMs). In this study, we found that CD9 physically links JAM-A to αvβ3 integrin and that the ternary complex of JAM-A, CD9 and αvβ3 integrin regulates angiogenic signalling by bFGF. As it turned out in subsequent studies (see below), the physical connection of JAM-A with integrins through tetraspanins seems to emerge as a common principle underlying JAM-A’s signalling function in various contexts. This paper also revealed a new function of JAM-A that is independent from its role in regulating apico-basal polarity.
Iden S†, Misselwitz S†, Peddibhotla SSD, Tuncay H, Rehder D, Gerke V, Robenek H, Suzuki A and Ebnet K (2012). aPKC phosphorylates JAM-A at Ser285 to promote cell contact maturation and tight junction formation. J. Cell Biol. 196: 623-639 (†equal contribution).
In this study, we have addressed the possibility that JAM-A, which interacts with the PAR – aPKC complex (PAR-3 – aPKC – PAR-6) through PAR-3 (see Selected Publication #2) is itself a target of aPKC. In fact, we found that aPKC phosphorylates JAM-A at Ser285 and that this phosphorylation is important for JAM-A’s role in establishing apico-basal polarity and regulating tight junction formation in epithelial cells. We also found that JAM-A phosphorylation coincides with the recruitment of the PAR – aPKC complex at primordial, spot-like adherens junctions (pAJs), strongly suggesting that JAM-A localized at pAJs first serves to recruit the PAR – aPKC complex to the correct subcellular location and subsequently is instrumentalized by the PAR – aPKC complex to regulate polarization and tight junction formation.
Iden S, Rehder D, August B, Suzuki A, Wolburg-Buchholz K, Wolburg H, Ohno S, Behrens J, Vestweber D and Ebnet K (2006). A distinct PAR complex physically associated with VE-cadherin in vertebrate endothelial cells. EMBO Rep. 7: 1239-1246.
In this paper, we made the surprising observation that VE-cadherin, which is localized at adherens junctions of endothelial cells, directly interacts with PAR-3 and assembles a cell polarity complex that is different from the PAR – aPKC complex localized at tight junctions of epithelial cells. These findings indicated that endothelial cells have developed mechanisms to regulate cell polarity in an endothelial cell-specific manner and that VE-cadherin has adapted its C-terminal end to contain a PDZ domain-binding motif that allow VE-cadherin to directly interact with PAR-3.
Rehder D, Iden S, Nasdala I, Wegener J, Vestweber D and Ebnet K (2006). Junctional adhesion molecule (JAM)-A participates in the formation of apico-basal polarity through different domains. Exp. Cell Res. 312: 3389-3403.
In this study, we have performed a structure-function analysis to identify the regions in JAM-A that are important for its function in regulating apico-basal polarity and tight junction formation in epithelial cells. From the observation made in this study we learned that JAM-A must a) must be able to dimerize, and b) must be able to interact with PDZ domain-containing scaffolding proteins to regulate polarity in epithelial cells. We also learned that depletion of the membrane-distal Ig domain makes a dominant-negative mutant of JAM-A, which provided an important information for later studies.
Gliki G, Ebnet K, Aurrand-Lions M, Imhof BA and Adams RH (2004). Spermatid differentiation requires the assembly of a cell polarity complex downstream of JAM-C. Nature 431: 320-324.
This study performed in the lab of Ralf Adams in London (UK, now in Münster, Germany) provided for the first time evidence for a more general role of JAM family proteins in the regulation of cell polarity. Inactivation of the JAM-C gene in mice, surprisingly, resulted in infertility in males. The reason for this infertility turned to be the inability of immature spermatids to polarize, which is necessary for spermatids to develop from a round cell shape to an elongated cell shape. This polarization step probably is mediated by a heterophilic interaction between JAM-C on round spermatids and JAM-B on Sertoli cells.
Ebnet K, Suzuki A, Ohno S and Vestweber D (2004). Junctional Adhesion Molecules (JAMs): More molecules with dual functions? J.Cell Sci. 117: 19-29.
In this review article we have summarized the current knowledge on the physiological functions of JAM family members. We highlight the emerging roles of JAMs as regulators of transient cell-cell interactions between leukocytes, platelets and endothelial cells during immune responses and as mediators of stable cell-cell interactions in multicellular tissues.
Ebnet K, Aurrand-Lions M, Kuhn A, Kiefer F, Butz S, Zander K, Meyer zu Brickwedde M-K, Suzuki A, Imhof BA and Vestweber D (2003). The junctional adhesion molecule (JAM) family members JAM-2 and JAM-3 associate with the cell polarity protein PAR-3: A possible role for JAMs in endothelial cell polarity. J.Cell Sci. 116: 3879-3891.
In this paper we have addressed the question if the JAM family members JAM-B and JAM-C also interact with PAR-3. Our assumption was based on the structural similarity of the three JAM family proteins. We found that – like JAM-A – JAM-B and JAM-C directly interact with PAR-3. Since JAM-B and JAM-C are expressed by various cell types including endothelial cells, our findings suggested that endothelial cell polarity is regulated by JAM-B and possibly JAM-C interactions with PAR-3.
Ebnet K, Suzuki A, Horikoshi Y, Hirose T, Meyer zu Brickwedde M-K, Ohno S and Vestweber D (2001). The cell polarity protein ASIP/PAR-3 directly associates with Junctional Adhesion Molecule (JAM). EMBO J. 20: 3738-3748.
This paper identifies the cell polarity protein PAR-3/ASIP as JAM-A binding partner. PAR-3 is part of the PAR-3 – aPKC – PAR-6 polarity protein complex which is highly conserved through evolution and which I conserved from C.elegans to vertebrates. Together with our cooperation partners in Yokohama (Japan), we biochemically characterized the interaction between JAM-A and PAR-3. As one intriguing observation of this paper we find that JAM-A is localized at so-called “primordial, spot-like adherens junction” (pAJs) or “puncta” during early stages of cell-cell contact formation, where it serves to recruit PAR-3. Our findings thus identified JAM-A as positional cue for the correct localization of the PAR-3 – aPKC – PAR-6 complex (PAR – aPKC complex) in vertebrate epithelial cells.
Ebnet K, Schulz CU, Meyer zu Brickwedde M-K, Pendl GG and Vestweber D (2000). Junctional adhesion molecule (JAM) interacts with the PDZ domain containing proteins AF-6 and ZO-1. J.Biol.Chem. 275: 27979-27988.
In this paper, we describe the identification of the two scaffolding poteins AF-6/afadin and ZO-1 as direct interaction partners for JAM-A. We also find that AF-6/afadin can recruit JAM-A to cell-cell contacts. These findings provided the first basis for the further characterization of JAM-A’s role as adhesion receptor at epithelial cell-cell contacts.


