P2: Modelling the developing gonad – organoid- and iPSC-based approaches

Stefan Schlatt, Centre of Reproductive Medicine and Andrology (CeRA) (Homepage)
Project summary
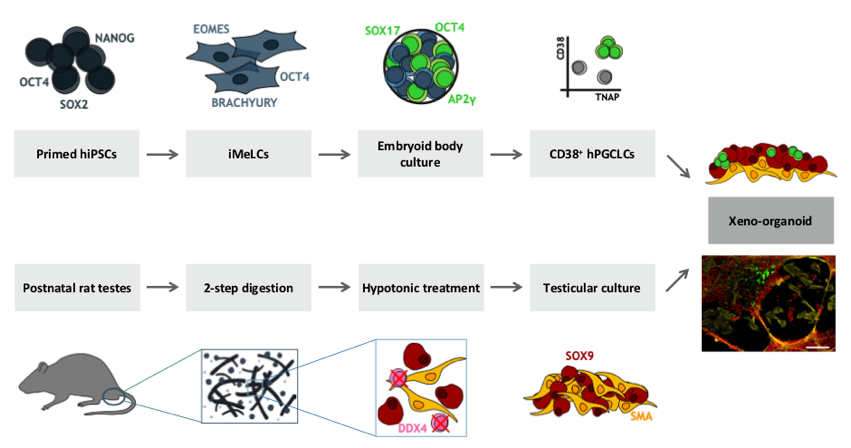
Human iPSC-derived primordial germ cell-like cells (hPGCLCs) currently represent the best experimental model to mimic early human germline development. In vivo, male fate is induced during testicular morphogenesis. Sertoli cells engulf germ cells and subsequently form aggregates as the first sign of seminiferous cord formation. Germ cells within seminiferous cords respond with male-specific transcriptional and epigenetic changes.
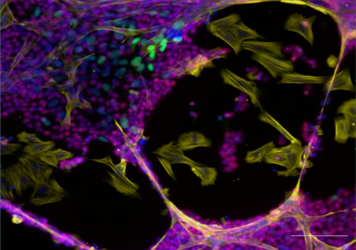
In the first funding period we established a xeno-organoid approach, consisting of reconstituted rat seminiferous tubule-like structures and co-cultured human PGCLCs. This enables us to describe crucial pathways involved in male somatic and germ cell specification. In this project we extend the experimental strategy. Xenografting of reconstituted testicular grafts and establishment of a homologous system (human primary testicular cells from adult transgender patients) opens fascinating scenarios for long-term and deep exploration of the influence of testicular microenvironments on fate decisions in human PGCLCs. Generation of human iPSC lines with loss-of-function (LoF) mutations in candidate genes via CRISPR/Cas will create options to correlate effects of gene knockouts on germline-somatic development. We expect that our findings help to identify pathways associated with impaired male fertility.