Theme and Objectives
Translational research:
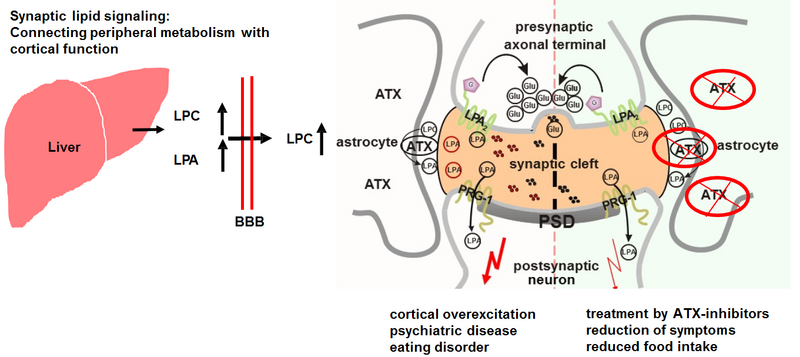
Our previous work using molecular and genetic approaches in model systems, but also human cohort data performed at the Institutes of Cell- and Neurobiology at the Charité-University Medical Center Berlin (from 1994-2009) and Microscopic Anatomy and Neurobiology at the University Medical Center of the Johannes Gutenberg-University Mainz (from 2010-2016) resulted in the discovery of a novel molecular pathway at the excitatory synapse in the brain. A role for bioactive lipid signaling at the synapse was first demonstrated by the detection of a new subgroup of lipid-phosphate phosphatases (LPPs)-like molecules, which we named plasticity related gene 1 (PRG-1, Bräuer et al., Nature Neuroscience, 2003). Unlike the classical LPPs, however, PRG-1 exhibits no phosphatase activity, but is a pore for lipid phosphatidic acid (LPA), a bioactive lipid acting on specific LPA1-6-receptors, thereby modulating excitatory transmission (Trimbuch et al., Cell 2009). This bioactive lipid signaling further involves the LPA-synthesizing molecule autotaxin (ATX) which is located in perisynaptic lamellae of astrocytes and converts lipid phosphatidic choline (LPC) to LPA in the synaptic cleft (Thalman et al., Molecular Psychiatry 2018). Dietary and pharmacological Intervention into this signaling pathway appears to be a novel therapeutic strategy to eventually treat psychiatric disease (Vogt et al., EMBO Mol Med 2015) and eating disorders. Building on these fundamental insights into the direct impact of peripheral metabolism on cortical function, clinical studies are on their way to develop these new therapeutic options to the patient’s benefit.
Philosophical studies:
Research on memory has been a major focus in the neurosciences over the past decades. An important advancement of memory research was achieved by Wilder Penfield at the Montreal Neurological Institute, who reported from the 1930s to the 1950s on experiential phenomena induced by electrical brain stimulation in humans, implying neuronal causation of memory. These reports as well as those on patient H. M. by Brenda Milner at the same institution continue to be referenced as ground-breaking, and neuroscientists have addressed the topic of memory from a range of sub-disciplines. Continuing experimental work by Nobel laureates Eric Kandel, John O’Keefe, as well as Edvard and May-Britt Moser related Penfield’s patient documentation to experiential phenomena. Assessing original charts of Penfield’s patients, however, questioned the stances that the neurosciences can uncover the “storehouse of memories”. Human memory must rather be regarded as context-sensitive and as an active reconstructive process, than a simple recording of events. Hence, strategies aiming at naturalizing all phenomena of mind (including memory) to cellular and molecular mechanisms cannot convincingly refer to Penfield’s electrophysiological studies, and remain unbacked but far-fetching claims to “understand man himself”. Further analyzes of such claims in the neuroscience literature are on their way.

