General
Endocytosis
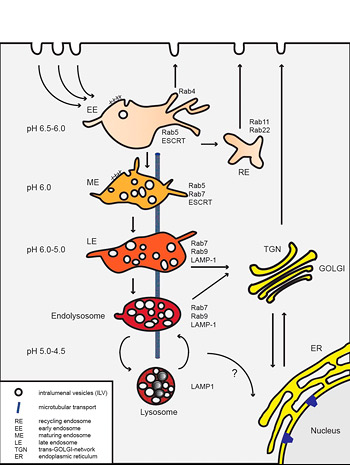
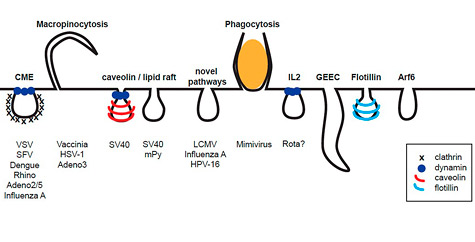
Endocytosis is an essential process of eukaryotic cells that is needed to internalize extracellular particles and solutes. Phagocytosis, the uptake of large particles, is typically restricted to specialized cells. In contrast, the uptake of small particles and solutes (pinocytosis) occurs virtually in all eukaryotic cells. For a long time it has been thought, that pinocytosis is mainly comprised of clathrin-mediated endocytosis. However, current research indicates a complex network of diverse ongoing and triggered pathways.
The pathways that viruses use for entry involve clathrin-coated vesicles, caveolae, macropinosomes, or further still poorly characterized pathways. Currently, we are particularly interested in a novel endocytic pathway used by Human Papillomaviruses type 16 (HPV-16) that bypass the classical organelles of established endocytic pathways.
Papillomaviruses
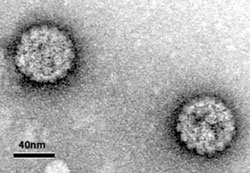
Die Erforschung des Infektionspotentials von Papillomviren ist offensichtlich für sich genommen wichtig. Eine Infektion mit bestimmten Papillomviren führt - neben dem Auftreten von Warzen in Haut und Schleimhautgewebe - zu tödlichen Gebärmutterhals- und anogenitalen Krebserkrankungen. Trotz Screening-Bemühungen und dem Aufkommen von Impfstrategien ist Gebärmutterhalskrebs immer noch die dritthäufigste Krebserkrankung bei Frauen weltweit und stellt daher weiterhin eine große Gesundheitsbedrohung dar, insbesondere in Entwicklungsländern.
Papillomviren sind kleine DNA-Viren, die aus zwei Strukturproteinen, L1 und L2, bestehen, aus denen die ikosaedrischen (T = 7) Kapside bestehen. Über den Eintritt der Papillomviren mit hohem Risiko ist wenig bekannt, hauptsächlich aufgrund der großen Schwierigkeiten, infektiöse Partikel in Gewebekultursystemen zu züchten. Für unsere Studien verwenden wir sogenannte Pseudovirionen, die infektiösen Partikeln ähneln, denen jedoch die virale DNA fehlt. Stattdessen enthalten die Partikel ein Reporterplasmid, das zur Überwachung eines effizienten endozytischen Eintritts verwendet wird.
Equipment

Neben modernen Labors und allen Geräten für Molekularbiologie, Biochemie und Virologie verwenden wir High-End-Mikroskopie-Ansätze wie Videomikroskopie, konfokale Laserscanmikroskopie, Totalreflexionsmikroskopie, konfokale Spinnscheibenmikroskopie, Elektronenmikroskopie und Elektronentomographie. Dafür arbeiten wir auf dem Campus und außerhalb des Campus zusammen, verwenden aber auch unsere eigene Ausrüstung, z. ein neues Spinning-Disk-Live-Cell-Mikroskop oder ein Totalreflexionsmikroskop.


