Our Research
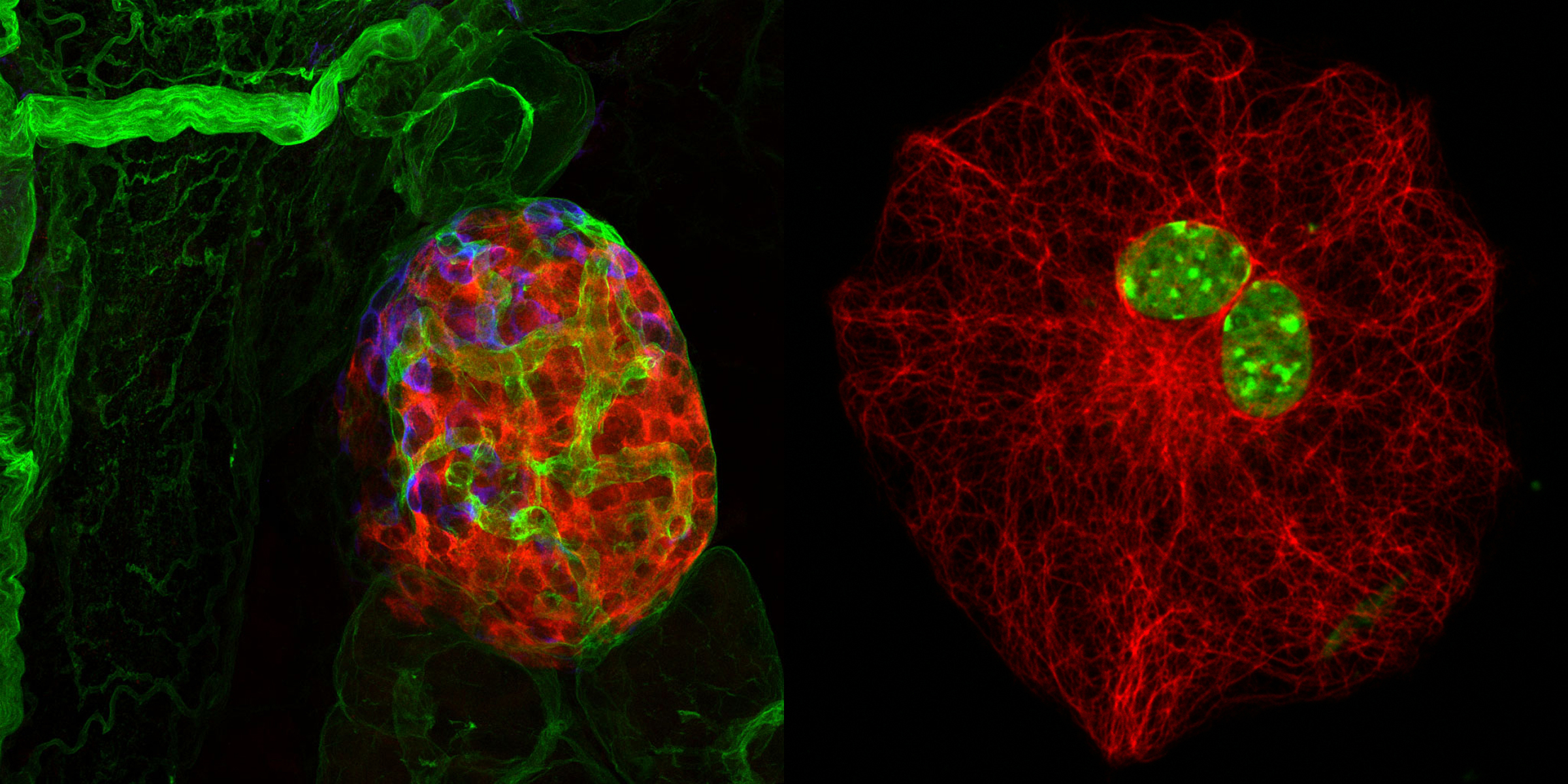
The focus of our group is blood vessels, inflammation and imaging, in particular of the brain, adipose tissue and secondary lymphoid organs, and how the extracellular matrix (ECM) affects the structure and function of these tissues and cellular processes within these tissues.
The ECM has different biochemical compositions and 3D structural forms that represent large portions of most tissues and vary depending on tissue type. We focus on thin sheet-like structures, known as basement membranes, that underlying polarized cells such as endothelial and epithelial cells, and ensheath muscle fibres, nerves and adipocytes.
It is well established that the ECM can influence the development, migration and survival of cells by direct receptor-mediated interactions. However, there is also evidence for indirect effects of the ECM resulting from presentation or controlled release of bound factors or bioactive fragments, and mechanical signals resulting from the 3D organization of the ECM. Our aim is to distinguish between the direct and indirect effects of the ECM, in particular basement membranes, on tissue development and homeostasis and how this affects inflammatory processes. We employ a multiscale imaging approach to study the temporo-spatial dynamics of cells within tissues, and to facilitate the transfer of our data to clinical application(Cells in Motion Interfaculty Centre).
Several models/ tissues are central to our investigations:
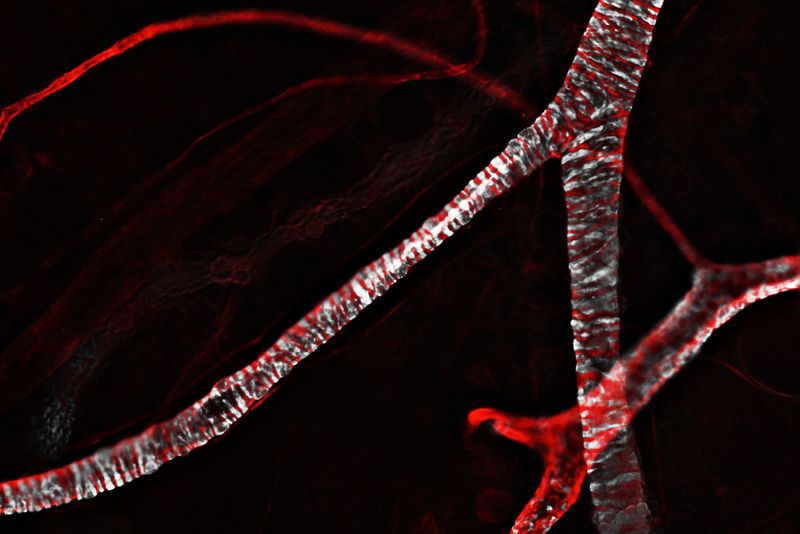
- Murine experimental autoimmune encephalomyelitis (EAE), a model for the human disease multiple sclerosis, in which we study how different leukocyte types transmigrate the postcapillary venules of the brain and the steps that are specific for brain blood vessels, with a focus on basement membranes and the perivascular space. Using novel transgenic mouse models we study the role of cell-matrix interactions in the localization and function of perivascular and CNS-resident myeloid cell populations (see TR CRC 128 and Marie Curie ITN ENTRAIN)
- To decipher the unique features of CNS blood vessels and neuroinflammation, comparisons are made between EAE and other inflammation models including type 1 diabetes (NOD mice), skin inflammation (delayed hypersensitivity - DTH), and cytokine- or pathogen-induced peritonitis and cremaster muscle leukocyte extravasation. To define routes and mechanisms of leukocyte infiltration into different peripheral tissues we employ live imaging to analyse the temporo-spatial dynamics of extravasating leukocytes (see CRC 1009; TR CRC 128 and Marie Curie ITN ENTRAIN).
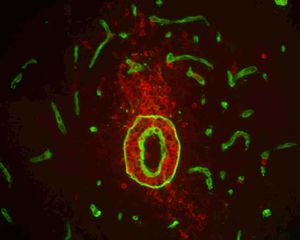
- A multiscale imaging approach is employed to analyse the temporo-spatial dynamics of extravasating myeloid populations into the omentum in sterile or pathogen induced peritonitis. Combined with single cell RNA sequencing of omental vessels and myeloid cells, we aim to identify marker molecules for the development of non-invasive imaging strategies for assessing acute peritoneal inflammation and/or for its manipulation (see CRC 1450 “inSight”, news item Research from cells to patients – Scientists make ongoing inflammation in the human brain visible)
- Murine models of stroke and small vessel disease are employed to investigate on the role of the ECM on structural and functional integrity of the neurovascular unit, and the role of inflammation and, in particular, myeloid cells in these pathologies (see svds-at-target).
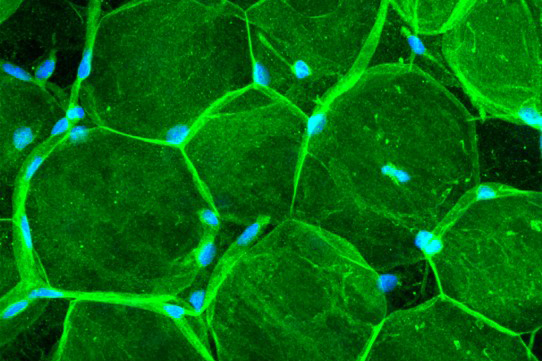
- Adipose tissues and obesity. Each adipocyte is encased in its individual basement membrane which we have characterized and shown to play a role in high fat diet-induced insulin resistance. Our studies focus on perivascular myeloid populations and their role in tissue homeostasis and obesity (German Research Foundation funded joint project with China, SO285/14-1).
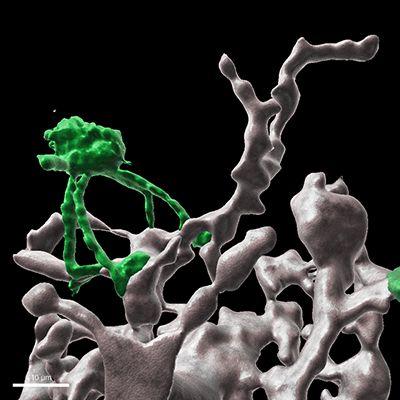
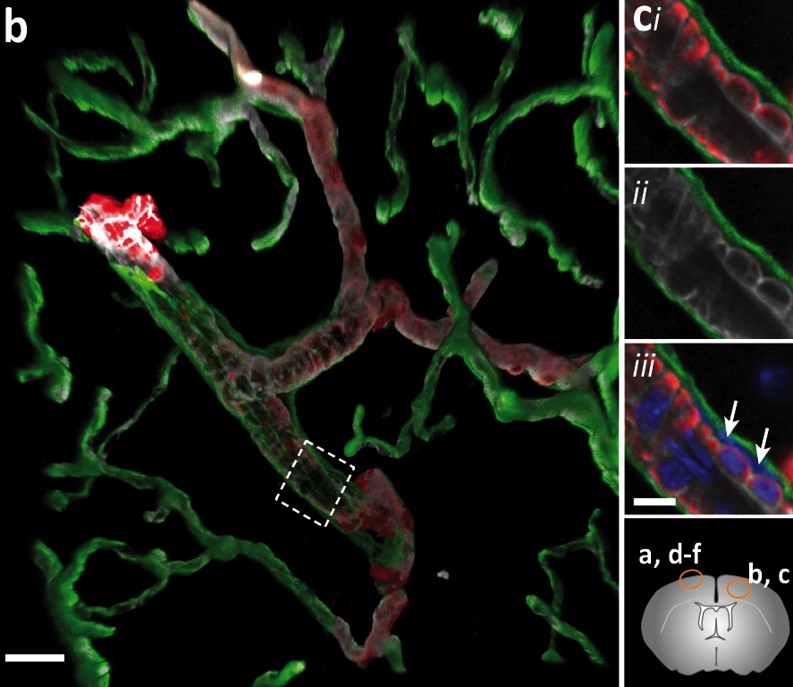
- Studies to date on leukocyte entry into the brain focus on endothelial properties or immune cell behaviour with little consideration of the 3D relationship between cellular and basement membrane barriers and their functional interdependence. In the Advanced ERC Grant project ‘B3M - Breaking into the Brain – Basement membranes and the perivascular niche’ we use new dextran-hydrogels to recreate not only the cellular barriers but also the basement membrane barriers which we know from our in vivo studies to play critical roles in constituting the blood-brain barrier. One focus of this project is the perivascular niche defined by the basal surface of the endothelium, the endothelial basement membrane (BM) and an outer parenchymal BM with associated astrocyte endfeet - a poorly studied site but of utmost fundamental and clinical relevance. We investigate the role of macrophages located at this perivascular site on cerebral microvessel function using split cre technology to target these cells. This is a cross-disciplinary project involving advanced in vitro hydrogel models and in vivo studies, aimed at deciphering cellular and molecular events occurring after leukocyte penetration of the endothelium, in the perivascular niche, required for final infiltration into the brain parenchyma.