Oligodendrocytes in health and disease

The Kuhlmann lab studies the molecular mechanisms of myelination and remyelination. The myelinating cells of the central nervous system (CNS) are oligodendrocytes. In many CNS diseases (e.g. multiple sclerosis (MS)) oligodendrocytes are destroyed resulting in demyelination and impaired neuronal function. Our research focuses on mechanisms regulating oligodendroglial cell death and remyelination, a repair process in the CNS. The aim of our research is to contribute to the development of new treatment strategies for demyelinating diseases.
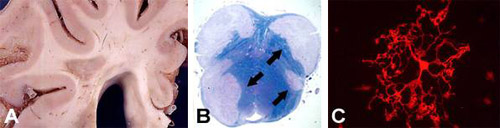
In current projects we analyze molecular mechanisms regulating oligodendroglial migration as well as the functional role of selected signalling cascades on oligodendroglial differentiation and remyelination. To address our research questions we use a wide range of experimental techniques and approaches. We study human tissue sections, primary rodent cell cultures, organotypic cerebellar slices and different demyelinating animal models.
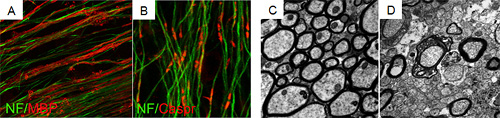
To translate our results from the rodent to the human system we established a protocol to generate oligodendrocytes from human induced pluripotent stem cells (iPSC). Using this technique, we study the phenotypes of oligodendrocytes from patients with different neurological diseases, dissect underlying molecular pathways and search for new treatment options.
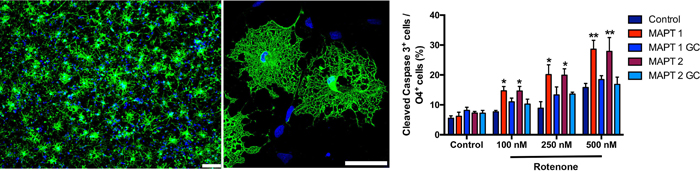
Our projects are funded by the DFG, SFB-TRR 128, International Progressive MS Alliance, National MS Society, The Marie Sklodowska-Curie Action of the European Union (H2020-MSCA-ITN-2018) and IZKF Münster.

Selected publications
- Starost L, Lindner M, Herold M, Xu YKT, Drexler HCA, Heß K, Ehrlich M, Ottoboni L, Ruffini F, Stehling M, Röpke A, Thomas C, Schöler HR, Antel J, Winkler J, Martino G§, Klotz L§, Kuhlmann§: Extrinsic immune cell-derived, but not intrinsic oligodendroglial factors contribute to oligodendroglial differentiation block in multiple sclerosis. Acta Neuropathol. 2020 140:715-736
- Heß K, Starost L, Kieran NW, Thomas C, Vincenten MCJ, Antel J, Martino G, Huitinga I, Healy L, Kuhlmann T: Stage dependent causes for remyelination failure in MS lesions. Acta Neuropathol. 2020 140:359-375.
- Weider M#, Starost LJ#, Groll K#, Küspert M, Sock E, Wedel M, Fröb F, Schmitt C, Baroti T, Hartwig A, Hillgärtner S, Piefke S, Fadler T, Ehrlich M, Ehlert C, Stehling M, Albrecht S, Jabali A, Schöler HR, Winkler J, Kuhlmann T*, Wegner M*. Nfat/calcineurin signaling promotes oligodendrocyte differentiation and myelination by transcription factor network tuning. Nat Commun. 2018 9: 899 doi: 10.1038/s41467-018-03336-3. # and * equal contribution.
- Stangel M, Kuhlmann T, Matthews PM, Kilpatrick TJ. Achievement and obstacles of remyelinating therapies in multiple sclerosis. Nat Rev Neurol. 2017:12:742-754. doi: 10.1038/nrneurol.2017.139.
- Ehrlich M, Mozafari S, Glatza M, Starost L, Velychko S, Hallmann AL, Cui QL, Schambach A, Kim KP, Bachelin C, Marteyn A, Hargus G, Johnson RM, Antel J, Sterneckert J, Zaehres H, Schöler HR, Baron-Van Evercooren A, Kuhlmann T. Rapid and efficient generation of oligodendrocytes from human induced pluripotent stem cells using transcription factors. PNAS 2017:114: E2243-E2252. doi: 10.1073/pnas.1614412114
- Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017 133:13-24.
- Preisner A*, Albrecht S*, Cui Q, Hucke S, Ghelman J, Hartmann C, Taketo MM, Antel J, Klotz L, Kuhlmann T. Non-steroidal anti-inflammatory drug indometacin enhances endogenous remyelination. Acta Neuropathol. 2015; 130: 247-61.* equal contribution
- Hargus G,*, Ehrlich M,*, Arauzo-Bravo MJ, Hallmann AL, Hemmer K, Reinhardt P, Kim KP, Adachi K, Santourlidis S, Ghanjati F, Kim JB, Sterneckert J, Schwamborn J, Schöler HR, Kuhlmann T#, Zaehres H#. Neural induction of human iPS cells reveals origin-dependent neural cell identities in vitro and after transplantation into the adult rodent brain. Cell Reports 2014; 8: 1697-703. *and # equal contribution.
- Goldschmidt T, Antel J, König F, Brück W*, Kuhlmann T*. The remyelination capacity of the MS brain decreases with disease chronicity. Neurology 2009, 72: 1914-21. * shared authorship
- Kuhlmann T, Miron V, Cui Q, Wegner C, Antel J, Brück W.. Maturation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic MS. Brain 2008, 131: 1749 -58
Contact:
Tanja Kuhlmann (Email)

