Prof. Dr. med. Tanja Kuhlmann

Institut für Neuropathologie
Pottkamp 2
Tel.: +49-(0)251-83-56968
Fax.: +49-(0)251-83-56971
Email
Research
Oligodendrocytes in health and disease
The Kuhlmann lab studies the molecular mechanisms of myelination and remyelination. The myelinating cells of the central nervous system (CNS) are oligodendrocytes. In many CNS diseases (e.g. multiple sclerosis (MS)) oligodendrocytes are destroyed resulting in demyelination and impaired neuronal function. Our research focuses on mechanisms regulating oligodendroglial cell death and remyelination, a repair process in the CNS. The aim of our research is to contribute to the development of new treatment strategies for demyelinating diseases.
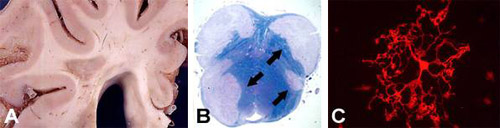
In current projects we analyze molecular mechanisms regulating oligodendroglial migration as well as the functional role of selected signalling cascades on oligodendroglial differentiation and remyelination. To address our research questions we use a wide range of experimental techniques and approaches. We study human tissue sections, primary rodent cell cultures, organotypic cerebellar slices and different demyelinating animal models.
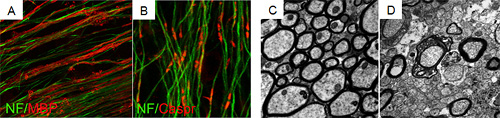
To translate our results from the rodent to the human system we established a protocol to generate oligodendrocytes from human induced pluripotent stem cells (iPSC). Using this technique, we study the phenotypes of oligodendrocytes from patients with different neurological diseases, dissect underlying molecular pathways and search for new treatment options.
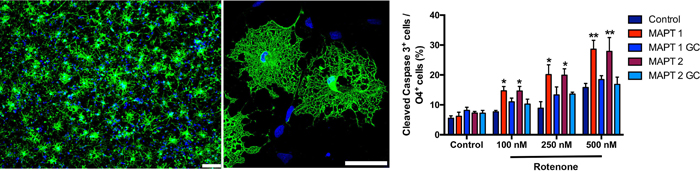
Our projects are funded by the DFG, SFB-TRR 128, International Progressive MS Alliance, National MS Society, The Marie Sklodowska-Curie Action of the European Union (H2020-MSCA-ITN-2018) and IZKF Münster.

Special model: Induced pluripotent stem cells
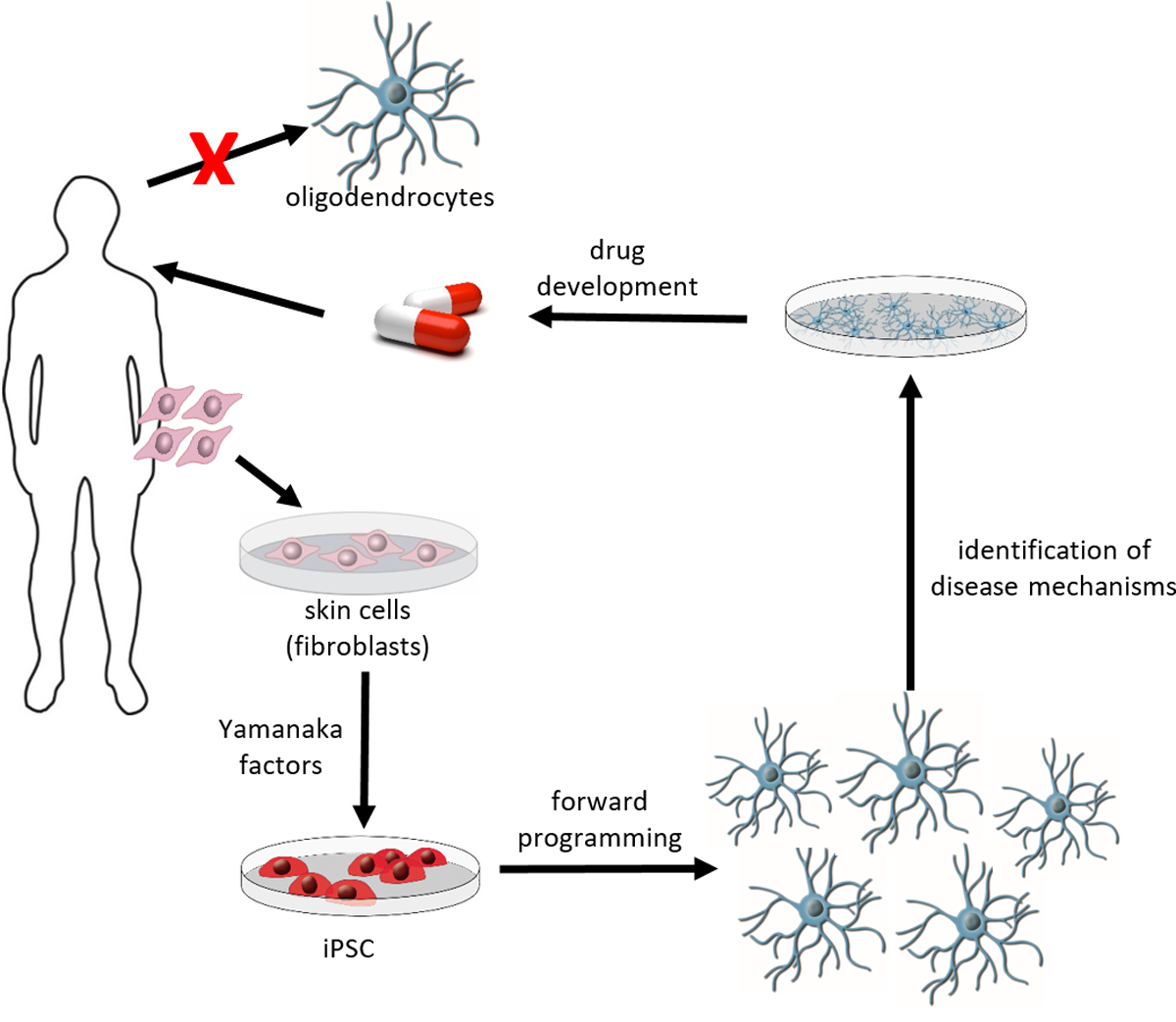
Diseases of the human central nervous system (CNS) represent a major challenge. The numbers of patients with diseases of the central nervous system are constantly increasing due to better health care and increasing life expectancy. Despite intensive research efforts in universities, research institutes and pharmaceutical industry the treatment options for many CNS diseases are still nonsatisfying. One reason for the limited success so far is the restricted availability of primary human CNS cells, such as neurons, astrocytes and oligodendrocytes. In 2006, a new technology was presented by Nobel Laureate Shinya Yamanaka to derive pluripotent stem cells from differentiated somatic cells without the need to isolate pre-implantation blastocysts. These induced pluripotent stem cells (iPSC) can be generated by introducing a set of pluripotency-associated transcription factors into differentiated somatic cells, which modify their epigenetic status leading to cellular reprogramming. As pluripotent cells, iPSC share main characteristics of embryonic stem cells, such as the ability for unlimited self-renewal and the potential to differentiate into cells of the three germ layers, mesoderm, endoderm and ectoderm. Since iPSC can readily be derived from human cells, including fibroblasts from skin biopsies, this technology has significant implications for regenerative medicine and biomedical research. Indeed, the derivation of iPSC from patients offers an invaluable opportunity to study disease pathogenesis in an in vitro-culture system on human cells at risk (e.g. oligodendrocytes or neurons from patients with myelin or neurodegenerative diseases), which is often difficult to accomplish in patients due to the unavailability of appropriate tissue (e.g. brain tissue of a patient).
Rapid, efficient and reproducible generation of human oligodendrocytes from iPSC
Oligodendrocytes are the myelin forming cells of the CNS and they play a key role in myelin related diseases including multiple sclerosis as well as neurodegenerative diseases or traumatic brain injury. Stem cell technologies, especially induced pluripotent stem cells are promising technologies to generate unlimited numbers of human oligodendrocytes. Access to large numbers of human oligodendrocytes will help to identify disease mechanisms and to develop new pharmaceutical treatment options. During recent years, we developed a protocol that allows the generation of human iPSC derived oligodendrocytes by forced expression of three transcription factors, namely SOX10, OLIG2 and NKX6.2 in iPSC derived neural progenitor cells (Ehrlich et al, PNAS 2017). Aim of this project is to optimize further the protocol to allow the fast, rapid and reliable production of human iPSC derived oligodendrocytes in high through put to facilitate and promote research of human CNS diseases and development of new treatment approaches in academia and pharmaceutical industry. This project is supported by the European Union and EFRE.NRW.
Selected publications
- Starost L, Lindner M, Herold M, Xu YKT, Drexler HCA, Heß K, Ehrlich M, Ottoboni L, Ruffini F, Stehling M, Röpke A, Thomas C, Schöler HR, Antel J, Winkler J, Martino G§, Klotz L§, Kuhlmann§: Extrinsic immune cell-derived, but not intrinsic oligodendroglial factors contribute to oligodendroglial differentiation block in multiple sclerosis. Acta Neuropathol. 2020 140:715-736
- Heß K, Starost L, Kieran NW, Thomas C, Vincenten MCJ, Antel J, Martino G, Huitinga I, Healy L, Kuhlmann T: Stage dependent causes for remyelination failure in MS lesions. Acta Neuropathol. 2020 140:359-375.
- Weider M#, Starost LJ#, Groll K#, Küspert M, Sock E, Wedel M, Fröb F, Schmitt C, Baroti T, Hartwig A, Hillgärtner S, Piefke S, Fadler T, Ehrlich M, Ehlert C, Stehling M, Albrecht S, Jabali A, Schöler HR, Winkler J, Kuhlmann T*, Wegner M*. Nfat/calcineurin signaling promotes oligodendrocyte differentiation and myelination by transcription factor network tuning. Nat Commun. 2018 9: 899 doi: 10.1038/s41467-018-03336-3. # and * equal contribution.
- Stangel M, Kuhlmann T, Matthews PM, Kilpatrick TJ. Achievement and obstacles of remyelinating therapies in multiple sclerosis. Nat Rev Neurol. 2017:12:742-754. doi: 10.1038/nrneurol.2017.139.
- Ehrlich M, Mozafari S, Glatza M, Starost L, Velychko S, Hallmann AL, Cui QL, Schambach A, Kim KP, Bachelin C, Marteyn A, Hargus G, Johnson RM, Antel J, Sterneckert J, Zaehres H, Schöler HR, Baron-Van Evercooren A, Kuhlmann T. Rapid and efficient generation of oligodendrocytes from human induced pluripotent stem cells using transcription factors. PNAS 2017:114: E2243-E2252. doi: 10.1073/pnas.1614412114
- Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017 133:13-24.
- Preisner A*, Albrecht S*, Cui Q, Hucke S, Ghelman J, Hartmann C, Taketo MM, Antel J, Klotz L, Kuhlmann T. Non-steroidal anti-inflammatory drug indometacin enhances endogenous remyelination. Acta Neuropathol. 2015; 130: 247-61.* equal contribution
- Hargus G,*, Ehrlich M,*, Arauzo-Bravo MJ, Hallmann AL, Hemmer K, Reinhardt P, Kim KP, Adachi K, Santourlidis S, Ghanjati F, Kim JB, Sterneckert J, Schwamborn J, Schöler HR, Kuhlmann T#, Zaehres H#. Neural induction of human iPS cells reveals origin-dependent neural cell identities in vitro and after transplantation into the adult rodent brain. Cell Reports 2014; 8: 1697-703. *and # equal contribution.
- Goldschmidt T, Antel J, König F, Brück W*, Kuhlmann T*. The remyelination capacity of the MS brain decreases with disease chronicity. Neurology 2009, 72: 1914-21. * shared authorship
- Kuhlmann T, Miron V, Cui Q, Wegner C, Antel J, Brück W.. Maturation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic MS. Brain 2008, 131: 1749 -58


