Differentiation and Maturation of Chondrocytes
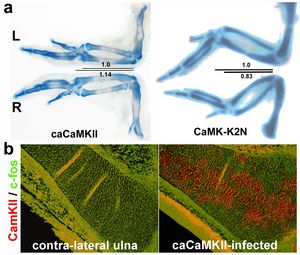
The various skeletal elements of the vertebrate skeleton differ in size and shape, but little is known about the molecular mechanisms controlling these two features. Nevertheless, perturbations in the chondrocyte maturation process lead to changes in the size of skeletal elements. Studying knock-out animals for Wnt9a we uncovered a very specific requirement for this Wnt-ligand during long-bone development: Wnt9a via β-catenin controls the expression of the central regulator of chondrocyte maturation, Indian hedgehog, in prehypertrophic chondrocytes in a spatio-temporal manner (Spaeter et al., 2006). Thus, providing a mechanism to fine-tune the size of the future skeletal element. Previously we have uncovered a potential role for Calcium/Calmodulin dependent kinase II (CaMKII) in the maturation process of chondrocytes in the chick (Taschner et al., 2008) (Figure 2). Yet, transgenic approaches in the mouse revealed only minor effects (Amara et al. 2017). The last step in endochondral ossification is the removal of hypertrophic chondrocytes and the formation of trabecular bone at the so-called chondro-osseous front. Here, we and others have shown that hypertrophic chondrocytes can take on an alternative fate and differentiate into osteoblasts participating in trabecular bone formation and that this process is positively influenced by the canonical Wnt pathway (Park et al. 2015; Houben et al. 2016).

